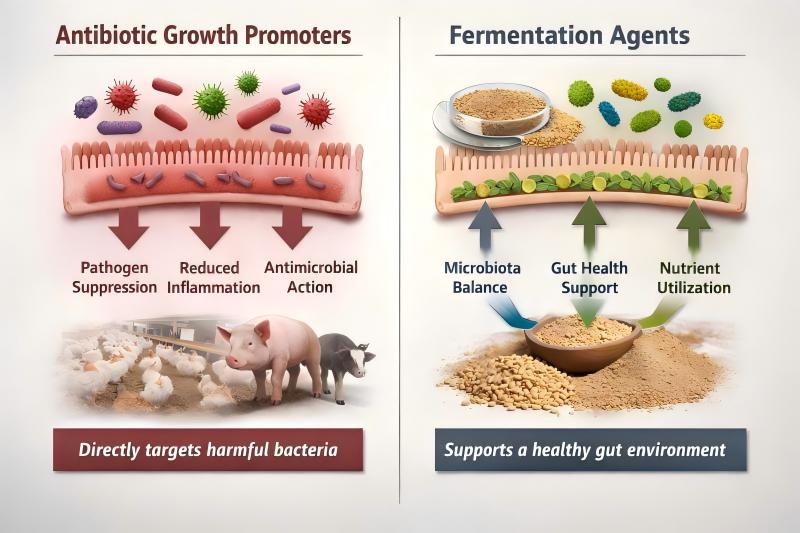
For decades, antibiotic growth promoters (AGPs) played a key role in stabilizing gut health, improving feed efficiency, and supporting consistent animal performance in intensive livestock systems.
However, as regulatory pressure increases and global producers accelerate the transition away from AGPs, the industry is actively searching for non-antibiotic growth promoter alternatives that can maintain productivity without compromising animal health.
Among the available options, fermentation agents—particularly yeast-based fermentation products—are frequently discussed.
But an important question remains: can fermentation agents truly replace antibiotic growth promoters, or do they serve a fundamentally different role?
Why Antibiotic Growth Promoters Worked So Well
Any realistic discussion about AGP alternatives must begin by acknowledging why antibiotics were effective.
Antibiotic growth promoters supported performance primarily by:
-
Suppressing subclinical pathogenic bacteria in the gut
-
Reducing chronic intestinal inflammation
-
Stabilizing nutrient absorption and feed conversion
-
Limiting performance losses under stress and high stocking density
These effects were especially evident in young animals and during periods of physiological stress.
Expecting fermentation agents to succeed without understanding these functions often leads to unrealistic expectations.
Why Fermentation Agents Are Considered AGP Alternatives
Fermentation agents operate through a different biological pathway than antibiotics.
Rather than directly killing bacteria, fermentation agents support gut function by:
-
Modulating the intestinal microbial ecosystem
-
Enhancing fiber and nutrient utilization
-
Supporting gut barrier integrity and immune balance
-
Creating a digestive environment less favorable to pathogenic overgrowth
In yeast-based fermentation agents, the fermentation process generates a range of functional metabolites, including organic acids, peptides, and yeast-derived components.
For example, certain fermentation products contain mannan oligosaccharides (MOS), which can bind specific pathogenic bacteria in the gut and reduce their ability to adhere to the intestinal wall.
This indirect mechanism helps stabilize gut health without antimicrobial action, which is why fermentation agents are commonly evaluated during the transition away from AGPs.

Can Fermentation Agents Fully Replace AGPs? The Real Answer
Short answer: Not in every situation.
More accurate answer: It depends on how “replacement” is defined.
Fermentation agents do not replicate the antimicrobial effects of antibiotics. Their effectiveness depends on:
-
Overall farm hygiene and biosecurity
-
Animal species and production stage
-
Feed formulation and ingredient consistency
-
The specific fermentation agent and its functional focus
-
Application strategy and duration
In well-managed systems, fermentation agents can support feed efficiency and gut stability at levels comparable to AGP-based programs.
In environments with high pathogen pressure, however, fermentation agents alone may not fully compensate for the loss of antimicrobial control.
Why Fermentation Agents Sometimes Fail to Deliver AGP-Like Results
Disappointing results are often attributed to “product inefficacy,” but the underlying causes are usually more practical.
Common issues include:
-
Selecting a fermentation agent that does not match the production goal
-
Using insufficient or inconsistent inclusion rates
-
Expecting immediate antimicrobial effects
-
Applying products for too short a trial period
-
Lack of technical guidance during implementation
These challenges are explored further in related analyses such as Why Yeast Culture Doesn’t Always Improve Feed Efficiency and Common Reasons Fermentation Products Fail in Livestock Diets.
In many cases, performance gaps reflect strategy misalignment rather than product failure.
When Fermentation Agents Can Effectively Replace AGPs
Fermentation agents perform best when used as part of a gut health–driven feeding strategy, not as a direct antibiotic substitute.
They are particularly effective in:
-
Antibiotic-free or reduced-antibiotic production programs
-
Systems focused on long-term feed efficiency rather than short-term growth acceleration
-
Stress-prone stages such as weaning, diet transitions, or environmental changes
-
Operations emphasizing gut resilience and microbial balance
In these contexts, Saccharomyces-based fermentation agents can support stable performance while reducing reliance on antibiotic growth promoters.
Product Selection Matters More Than Performance Claims
One of the most critical—and often overlooked—factors in AGP replacement programs is product selection.
Not all fermentation agents serve the same function.
For example:
-
Fermentation agents intended to support hindgut stability and pathogenic bacteria control require specific strain selection and metabolite profiles, often emphasizing yeast cell wall components such as MOS.
-
In contrast, products designed to improve rumen fiber digestion focus on entirely different fermentation characteristics and functional metabolites.
Using a fermentation agent designed for fiber utilization to address pathogenic pressure is unlikely to deliver meaningful results.
This functional mismatch explains why many AGP replacement attempts underperform.
The key takeaway is simple: selection logic matters more than marketing claims.
Choosing Fermentation Agents for AGP-Free Programs
When evaluating fermentation agents as non-antibiotic growth promoter alternatives, decision-makers should focus on:
-
Functional objective (gut stability, feed efficiency, stress mitigation)
-
Species-specific requirements
-
Fermentation strain origin and process control
-
Consistency and quality assurance
-
Availability of technical support for application optimization
Solutions such as Saccharomyces Brewing Yeast Culture, including general-use and ruminant-specific formulations, illustrate how fermentation agents can be tailored to different production goals rather than marketed as universal replacements.
Conclusion: Rethinking Replacement in the Transition Away from AGPs
Replacing antibiotic growth promoters is not about finding a one-to-one substitute.
It is about redesigning feeding strategies to support gut health, nutrient utilization, and long-term system stability.
Fermentation agents can play a meaningful role in this transition—when they are correctly selected, properly applied, and integrated into a broader nutritional framework.
If you are currently evaluating fermentation-based solutions as part of your transition away from AGPs, a targeted technical assessment can help determine which approach best fits your production system.
FAQs
Q1. Can fermentation agents completely replace antibiotic growth promoters?
Fermentation agents cannot fully replace antibiotic growth promoters in every production system. They do not provide direct antimicrobial effects, but can support gut health, nutrient utilization, and performance stability when properly selected and applied, especially in well-managed, antibiotic-free programs.
Q2. Why are fermentation agents considered non-antibiotic growth promoter alternatives?
Fermentation agents support animal performance through gut microbiota modulation, improved digestion, and functional metabolites rather than bacterial suppression. This makes them suitable as non-antibiotic growth promoter alternatives during the transition away from AGPs.
Q3. What functional metabolites make fermentation agents effective?
Yeast-based fermentation agents may contain functional metabolites such as organic acids, peptides, and mannan oligosaccharides (MOS). MOS can help bind specific pathogenic bacteria and reduce gut colonization without antimicrobial action.
Q4. Why do fermentation agents sometimes fail to replace AGPs?
Common reasons include incorrect product selection, mismatched functional goals, insufficient dosage, short application periods, and unrealistic expectations of antimicrobial effects. Performance issues are often strategy-related rather than product-related.
Q5. How should fermentation agents be selected for AGP-free programs?
Selection should be based on production goals such as gut stability, feed efficiency, or fiber digestion. Products targeting hindgut health differ fundamentally from those designed for rumen fiber utilization, making functional alignment more important than performance claims.
