Introduction
Inconsistent results are one of the most common concerns when using fermentation-based feed additives. The same product may deliver strong performance in one batch, but noticeably weaker results in another.
While many factors can influence outcomes, one critical element is often overlooked: microbial stability in fermentation products.
Without stable microbial activity and consistent metabolite profiles, even well-formulated products can fail to deliver reliable performance. Understanding this factor is essential for both technical teams and purchasing decision-makers.
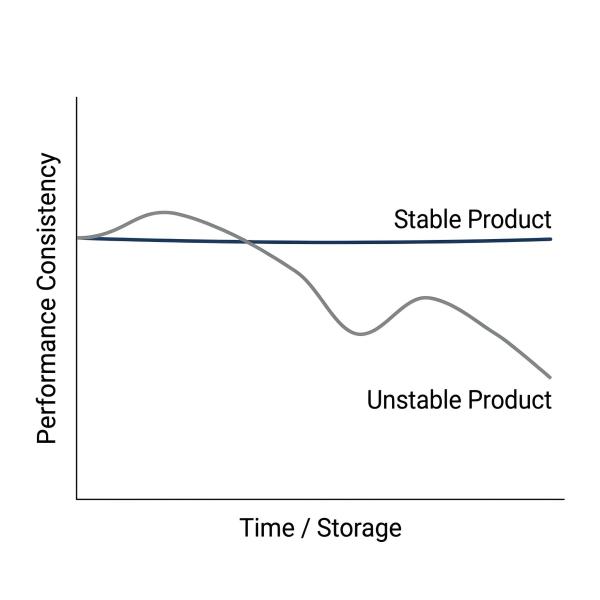
What Is Microbial Stability in Fermentation Products?
Microbial stability refers to the ability of a fermentation product to maintain consistent microbial viability, metabolic activity, and composition over time. In practical applications, this is especially important for products such as Saccharomyces cerevisiae culture , where functional performance depends not only on microbial presence, but on sustained biological activity throughout storage and use.
It includes three key dimensions:
- Viability – the survival of beneficial microorganisms
- Activity – the ability of microbes to produce functional metabolites
- Consistency – minimal variation between production batches
Why Microbial Stability Directly Affects Product Performance
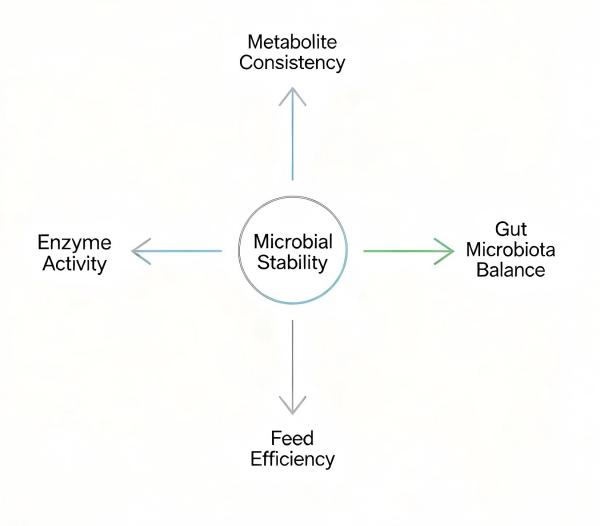
Impact on Metabolite Production
Fermentation products deliver value primarily through metabolites such as enzymes, organic acids, and bioactive compounds.
When microbial stability is compromised:
- metabolite levels fluctuate
- functional compounds degrade
- performance becomes inconsistent
This is also why understanding what defines a high-quality Saccharomyces cerevisiae culture is essential when evaluating fermentation-based feed additives.
Influence on Gut Microbial Balance
Stable fermentation products help support a balanced gut microbiota by:
- promoting beneficial bacteria
- suppressing harmful microorganisms
- maintaining digestive stability
Unstable products, however, may fail to consistently support these functions, reducing their effectiveness in real-world conditions.
Effects on Feed Efficiency and Animal Response
Ultimately, microbial stability impacts:
- nutrient digestibility
- feed conversion efficiency
- animal growth and productivity
Even small variations in microbial activity can translate into measurable differences in performance outcomes.
Key Factors That Affect Microbial Stability
Raw Material Quality
The fermentation process starts with raw materials. Variations or contamination at this stage can lead to:
- inconsistent microbial growth
- unstable fermentation outcomes
Fermentation Process Control
This is one of the most critical factors. For example, professionally developed fermentation agents rely on precise control of microbial environments to ensure stable metabolite production and consistent product quality.
Key variables include:
- temperature control
- fermentation duration
- pH management
Poor control can result in uneven microbial populations and unstable metabolite production.
Drying and Post-Processing
High temperatures or improper drying techniques can:
- damage microbial cells
- reduce biological activity
- degrade functional compounds
This stage often determines whether a product maintains its effectiveness after production.
Storage and Transportation Conditions
Even well-produced products can lose stability if not handled properly.
Key risks include:
- high humidity
- temperature fluctuations
- inadequate packaging
These factors directly affect shelf life and performance consistency.
Signs of Poor Microbial Stability in Fermentation Products
Identifying unstable products early can prevent performance losses.
Common indicators include:
- inconsistent results across batches
- noticeable changes in odor or appearance
- reduced shelf life
- clumping or physical degradation
These issues are often linked to unstable fermentation quality, rather than formulation alone.
How to Evaluate Microbial Stability Before Purchasing
For distributors and feed manufacturers, evaluating stability is essential.
Key checkpoints include:
- Batch consistency data – are results stable across different production lots?
- Quality control systems – does the supplier control fermentation conditions precisely?
- Stability testing – are shelf-life and activity verified over time?
- Technical support – can the supplier explain performance mechanisms clearly?
Choosing a supplier with strong production control and technical transparency significantly reduces risk.
Conclusion
Microbial stability is not a secondary characteristic—it is a core determinant of fermentation product performance.
Without it, even high-quality formulations may fail to deliver consistent results. With it, fermentation products can reliably support animal health, feed efficiency, and production outcomes.
For long-term success, focusing on consistency, process control, and supplier reliability is just as important as selecting the right product type.
